Preclinical Safety
In preclinical studies, direct lung delivery of a ThyrOxy™ dose 30-fold higher than the clinical trial dose resulted in no local lung injury or inflammation and no systemic side effects or clinical pathology.
Pharmacokinetics of Directly Instilled T3
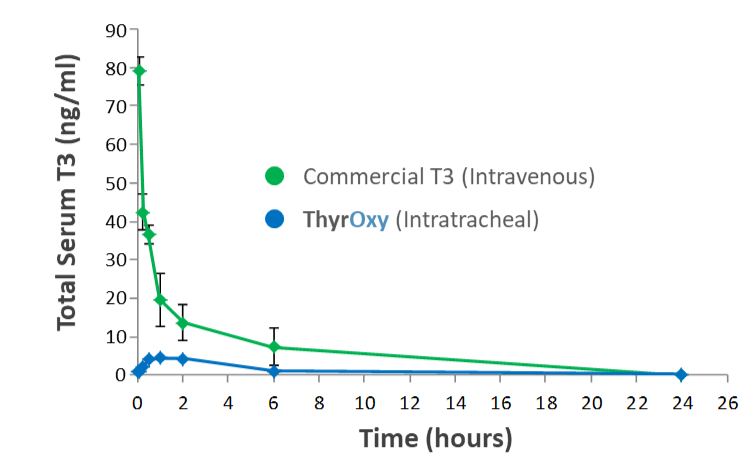
Direct lung delivery of ThyrOxy resulted in blood T3 concentrations that were 17X lower than those observed after intravenous administration. This significantly limits the potential for systemic side effects when ThyrOxy is delivered directly to the lungs.
Blood T3 After Intravenous and Direct Lung Delivery
ThyrOxy Safety Demonstrated in Preclinical Studies
Although direct lung delivery of intravenous T3 formulations led to rapid mortality, lung-delivered ThyrOxy demonstrated a strong safety profile. In a GLP-compliant IND-enabling study, ThyrOxy was delivered directly to the lungs on five consecutive days at the maximum feasible dose. This regimen resulted in no adverse clinical findings, clinical pathology, or signs of local lung injury or inflammation. (Triiodo-L-thyronine Rapidly Stimulates Alveolar Fluid Clearance in Normal and Hyperoxia-injured Lungs). Learn more about our research.

